




The electroless plating (EP) covered in this book is somewhat different from the original usage of the term by Brenner. Brenner’s use of the term electroless plating originally meant autocatalytic plating [1] . The term electroless plating, as used in this book, is not limited to the autocatalytic deposition but includes all chemical plating. So, the defi nition of electroless plating, as used in this book,involves immersing a substrate/workpiece into a bath solution, a metal/alloy or compound layer is then deposited on it via the transfer of electrons between reacting chemical species during the chemical reactions (redox reaction) without any applied current. It should be pointed out that displacement deposition is included in the electroless plating [2–4] . Displacement deposition is also known as galvanic or immersion plating. A more noble metal, that is the redox potential of a metal ion in solution, because it is more positive always deposits on the surface of a less noble metal in the galvanic displacement deposition. The less noble metal acts as a reducing agent. The difference between electroless plating and galvanic displacement deposition is that a reducing agent is necessary in former case, but is not required in latter case. That is why galvanic displacement deposition is simpler and faster [5–8] .
There are a lot of advantages of EP compared to traditional electrolytic deposition, such as:
No external electric source, including rectifi ers, batteries or anodes is needed so that the equipment needed for electroless plating is rather simple.
Excellent uniformity and less porous deposits are produced, even on complex components. On the other hand, there exists the phenomenon of current density concentration at peaks and protrusions in the electrolytic plating process. So, the fi lms produced by electrolytic plating are not always uniform,there are nearly no deposits at protrusions or in the ecesses. In addition,the fi lms always have pinholes, which make the deposits susceptible to rust.
The electroless deposition process is easy to use in volume production.
Deposits can be produced directly on non-conductive materials such as ceramics and plastics and other so-called hard plating substrates.
Deposits usually have unique properties, which have applications in many industries.
Compared to the traditional electrolytic deposition process, there are also some drawbacks to electroless plating:
The EP baths are usually rather more complex than electrolytic deposition baths, so they are more expensive than electrolytic deposition.
Compared with the electrolytic plating process, EP has a limited life, and therefore the composition of consumption in the bath should be often added,especially during the process of the bulk production.
Electroless processes result in a higher waste treatment burden than that of electrolytic processes, which causes more troubles of waste solution treatment.
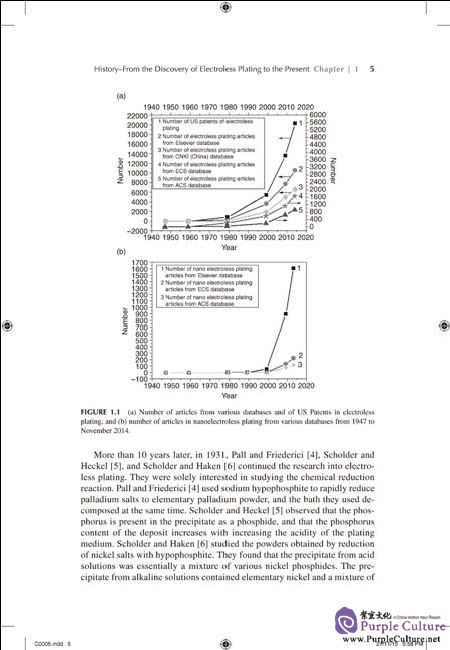
Since electroless plating was discovered by Brenner and Riddell in 1946,nearly seventy years have passed. EP is highly developed today, and is used in most parts of industry. Many basic subjects of EP, such as the improvement of bath solutions and new bath solutions, new alloy deposition, the extension of plating substrates, the mechanisms, especially the theory of electroless plating,nano electroless deposition, properties of in-depth study, and so forth have been studied and gratifying progress has been made. Considerable advances in such issues have been made in recent years, especially since the beginning of the new century. This can be corroborated from the fact that many papers were published in recent years. If you put “electroless plating” into the Elsevier data base, as of November 1, 2014, 10,565 papers can be found. The distribution of these articles is that 309 was published up to 1979; 3,323 from 1980 to 1999; 4,230 from 2000 to 2009; and 2,823 from 2009 to November 1, 2014. If you search within the results using “nano plating”, then 1,610 articles can be found up to January 23,2013, of which only 1 article was published up to 1979; another one was published between 1980 and 1989; 55 from 1990 to 1999; 885 from 2000 to 2009;and 660 from 2010 to January 23, 2013. That is to say, only a few papers on nano electroless plating were published in various journals included in the Elsevier data base before 2000, and most of them were been published after 2001.
Few books on electroless plating have been published, for example, < Electroless plating: fundamentals and applications > by G. O. Mallory and J. B.Hajdu in 1990, and < Electroless nickel plating > by Wolfgang Riedel in 1991.It is obvious that these books can’t include recent developments in the materials used in electroless plating because they were published twenty years ago. Some books in electroless plating have been published in Chinese in recent years, in which nearly no nano electroless plating knowledge is included and not too many English papers in electroless plating were cited in any of them. One can see that there is an obvious gap in the existing literature that needs to be fi lled.It hoped that this book will fi ll the gap in knowledge left by existing electroless books.
Materials scientists, after studying the subject carefully enough, can streamline a thick metallurgy book into only few words: constituents, structures and properties; how to make steel or alloy material and their applications are not usually included in the contents of a metallurgy book. Similarly, a book on electroless plating can be streamlined into only few words as well: technology (manufacture process), constituents, structures and properties. Usually, how to manufacture electroless plating alloys is closely related to the contents of a book in electroless plating. Most electroless deposition books also include applications of electroless plating alloys. However in order to concentrate the contents of this book in a similar way to a common metallurgy book, and to avoid a lengthy of book, the properties and applications of EP will be not included in this book. In this way, this book will cover manufacturing technology plus the metallurgy of electroless plating alloys. In addition, in order to concentrate the fundamental content and theory of electroless plating, the subjects of pretreatment for electroless plating, post-treatment of electroless deposits,quality control of electroless deposits, stripping of electroless coating, treatment of waste bath solution, and equipment for electroless plating, which are usually included in other electroless plating books will not be included in this book.
There are four parts included in the book: 1, the history of electroless plating;2, the technology and manufacturing principles of electroless plating: plating bath, affecting parameters, deposition rate and stability of plating bath; 3,the composition, microscopic structure and surface morphology of electroless deposits; and 4, the principles of the formation, mechanism and theory of electroless plating.
Regarding the contents of the four parts of this book, we would like to say more as follows.
The history of electroless plating is narrated in detail in Part 1. It can be divided into several periods: the discovery of electroless plating, the early preparation stage (1946–1959), the slow stage of development (1960–1979),the rapid development and expansion stage (1980–1999), and the stage of nano electroless plating and the deep investigation of the mechanisms and theory of electroless plating (2000–). No electroless plating books have narrated and discussed the whole history of electroless plating in detail, and this is the fi rst time it has been done. The aim is to understand the development process of electroless plating more clearly and properly.
The part on technology includes the subjects of plating baths, affecting parameters,deposition rate, and the stability of plating baths. The aim in this part is not only to describe how to get EP alloy deposits, but also to sort and analyze the huge amount of experimental data and try to fi nd out what is a unifi ed or a regular kernel from the plentiful data, as well as seeking possible explanations. In addition,this part tries to identify the existence of possible confl icting phenomena in the experimental facts, and point out what questions need to be solved. There are three chapters on new subjects (Nano EP, EP Fe based alloy deposits, and Green EP) which appear in this part, and which will be illustrated below.
Some scholars describe electroless nickel plating (EN) as electroless plating.The reason for this is that EN is ndoubtedly of the great importance, and has been widely employed in various industries due to its unique physical and chemical properties. However, we think that electroless plating of Fe based alloys should be studied as well, even just from the point of view of pure basic investigation.That is why our group studied the electroless plating of Fe based alloys early on,from the middle of 1980s, and the study subjects of three graduate students in our group were all based on electroless plating in Fe based alloys. Therefore, a chapter on electroless plating in Fe based alloys is included in this part.
As mentioned above, there are nearly no papers on nano EP published before the new century and this area has not been included in nearly all existing electroless plating books. A chapter on nano electroless plating has therefore been included in this part as well.
People have realized that conventional electroless plating is not exactly a “green” technology because many chemicals are added into electroless bath solutions which may be toxic, for example,formaldehyde which is a known carcinogen. Therefore, how to make the technology of electroless plating green technology and to make the process more environmentally friendly are very important. Fortunately, scientists have begun to study the green technology of electroless plating and to make appropriate advances. So, a chapter on the green technology of electroless plating is included in this part, which represents another feature of the book.
In order to narrate more clearly, the baths of electroless plating has been classifi ed according to metals, binary, ternary, quaternary, quinary and multi alloys, but not to the various based alloys as is customary. However, the classification of the bath of electroless plating in binary alloys is still according to various based alloys as usual. Composite electroless deposits are discussed in a subsequent chapter in order to detail their nature. Many papers have reported the results of electroless plating on various substrates including on the so-called hard-coated substrates in order to enhance the applications of electroless plating and to better master the skills of fabrication. However, this subject will be not included in this book because it mainly represents the technical aspects of the subject (which of course is not to say that they are not important).
The plating rate and various affecting parameters are one of the main subjects in EP technology. Many EP papers concentrate on or refer to this subject,so the experimental data in the fi eld are very plentiful. It should be said that nearly no books or review papers concentrate on the description and analysis of these, especially no one has compared them in order to find universal phenomena and laws, to find problems such as the existence of conflict phenomenon, and to point out what needs to be done in the future. A special Chapter is included,and all of these points have been discussed.
The compositions and microstructures of electroless deposits are very important.Clarifying them not only helps in understanding the nature of EP deeply,but also closely connects with the properties and applications of EP deposits.Collecting the SEM micrographs of electroless alloy depositions together and concentrating discussion of them, people may understand the nature of electroless plating deposition more clearly and deeply. The contents of these two aspects are dispersed in many literatures, and no one has collected them together,analyzing and discussing them. That is why we use a part to describe and discuss them.
The part of kinetics, theory and mechanism of electroless plating includes the formation process of electroless deposits, their formation mechanism and the theory describing the formation of deposition. There has been new progress especially in the theory of describing the formation, such as the formation theory of EP amorphous alloys and EP microscopic theory, and these areas are generally no well-covered in existing electroless plating books. Here we pay particular attention to them.
The development of electroless plating in China was not started until the early 1980s because of the delay caused by the well-known Cultural Revolution.Our group was one of the fi rst institutions to study electroless plating in China. Except for the above-mentioned electroless plating of Fe based alloys,we studied some new electroless plating alloy systems, e.g. Cu-Ni-Sn-P quaternary alloy system, and the systematical study of some properties, especially the welding property of such alloy systems. Although the data and results of our research are included in the book, however they form only a small part of the whole book. Most of the results and data cited in the book are from scientifi coriginal papers published by various groups, companies and other institutions all over the world. More than 2,100 papers, reports and patents are cited in the book. Thereby, three points must be pointed out.
First, there is a huge amount of literature on electroless plating, only those closely related to the contents of the book were cited, others may not be referred to, which of course does not mean that these works are not important or not significant.
Second, the author, of course, wishes to seek proper use of the information cited. However, I don’t know if this is the case, and only the readers can make the right judgments.
Third, the author usually analyzes and evaluates the data and results cited,and may say some words about them. Of course, I don’t know if it is real correct and appropriate, so may I ask the readers to make their evaluation and comment.
At the beginning in designing this book, and during the whole writing process,I set a rule for myself that this book must not only well organize the cited results and data, providing a complete knowledge of EP to the readers, but also to analyze them and review them in as detail as possible. For the existed data,besides the achievements and progress, the possible problems and shortcomings must be boldly affirmed and pointed out, and the context and possible development for every subject discussed in the book also must be mentioned. In this way, the author hopes that readers can have an up-to-date understanding of the development process of EP, the basic academic subjects and the nature of EP from the book, thereby they not only can learn and master the existing knowledge of EP, but also can get some inspiration and enlightenment, and begin to think, consider and propose some new problems in the development of EP. This is my highest aim and greatest wish in writing this book. Now, the book is fi nished, I really do not know whether my rule has been xecuted, and my hope has been achieved. Only the readers can give right judgment about it.
I have pointed out many times in the book that after the discovery of EP nearly seventy year’s ago, great and plentiful achievements have been presented,which have come from the hard investigation of scientists all over the world. Until today, research in this fi eld has continued. There is no doubt to be expected that more and more new and deeper results will appear.
The materials contained within this book from our group were the crystallization of the collective efforts of the whole group in the fi eld of electroless plating research over many years. I wish to express my sincere thanks to all those who participated in the research, including my colleagues, my former graduate students and even many undergraduate students of mine. I’d like to list some but not all of their names here: Dr. Zhang Heng, Dr. Tang Zhaosheng,Dr. Yi Ge, Professor Dr. Hu Wangyu, Professor Dr. Liao Shuzhi, Professor Dr. Xie Haowen, Professor Dr. Shu Xiaolin, Professor Dr. Wang Lingling, and Professor Dr. Ouyang Yifang. I appreciate very much the Max-Plank-Institut für Plasmaphysik (IPP), Germany where I worked for a long period of time, for granting me the Max-Plank Scholarship, which enabled me to cooperate with Dr. E. Taglauer, and Dr. S. Lindig from IPP, in the study of the surface morphology of electroless deposits on Ni-P based alloy systems.
My friend Professor Dr. W. A. Jesser, University of Virginia, spent a lot of time in critically reading part of the manuscript, I am very grateful to him for his selfl ess help.
The writing of the book took more than four years. My wife has accompanied me day and night, so that I could concentrate on the writing and never feel alone. I really appreciate her help.
I express my thanks to the network center and library in Hunan University for their helping me in searching the relevant data. Particular thanks are due to Mrs. Xiao Wei and Mrs. Huang Xiaoling for helping me to reprint many papers that were diffi cult to get hold of. Also thanks are due to Mr. Hu Hao for helping me to use the network more smoothly. Many friends and relatives gave me both tangible and intangible help for many years, I express sincerely my gratitude to them.
Zhang Bangwei
Yuelu Mountain, Hunan University, New Year’s Day, 2015
REFERENCES
[1] Brenner A . P&SF 1984 ; 71 : 24 .
[2] Mallory O , Hajdu JB . Electroless Plating: Fundamentals and Applications . AESFS: Orlando,FL ; 1990 .
[3] Carraro C , Maboudian R , Magagnin L . Surf Sci Rep 2007 ; 62 : 499 .
[4] Djokic′ SS , Cavallotti PL . In: Djokic′ SS , editor. Electroless Deposition: Theory and Applications,in Modern Aspects of Electrochemistry , 48 . Springer ; 2010 . p. 251 – 89 .
[5] Yae S , Nasu N , Matsumoto K , Hagihara T , Fukumuro N , Matsuda H . Electrochim Acta2007 ; 53 : 35 .
[6] Lu Xianmao, Chen Jingyi, Skrabalak S E, Xia Younan, Proc. IMechE Vol. 221 Part N: J. Nanoengineering and Nanosystems 2008:1.
[7] Qiu T , Wu XL , Mei YF , Chu PK , Siu. GG . Appl Phys 2005 ; A 81 : 669 .
[8] Chong Hyun , Rheema Youngwoo , ChoaYong-Ho , Park Deok-Yong , Myung NV . Electrochim Acta 2010 ; 55 : 1072 .